Antibody (Suitable for clinical applications)
Sample Type: FFPE Patient Samples.
Tested Applications: IHC. Approved for In Vitro Diagnostic Procedures on FFPE tissues. For tissue collection recommendations, please see datasheet sent with product.
Application Notes
| Specification | Recommendation |
|---|---|
| Recommended Dilution (Conc) | 1:25-1:50 |
| Pretreatment | Citrate Buffer pH 6.0 |
| Incubation Parameters | 30 min at Room Temperature |
Prior to use, inspect vial for the presence of any precipitate or other unusual physical properties. These can indicate that the antibody has degraded and is no longer suitable for patient samples. Please run positive and negative controls simultaneously with all patient samples to account and control for errors in laboratory procedure. Use of methods or materials not recommended by enQuire Bio including change to dilution range and detection system should be routinely validated by the user.
Clonality: Monoclonal
Anti-Cytokeratin 10 Antibody Clone: DE-K10
Host and Isotype: Mouse IgG1, kappa
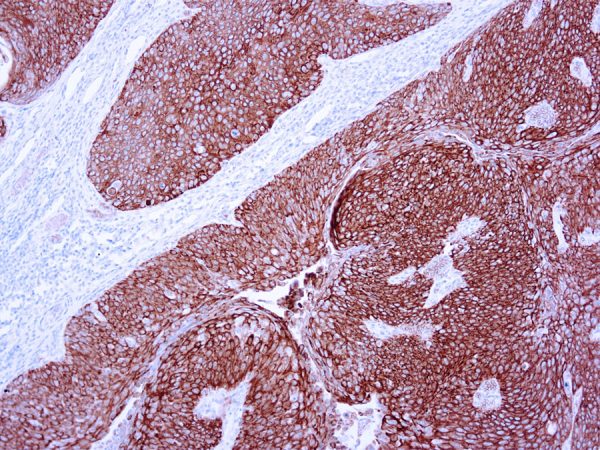
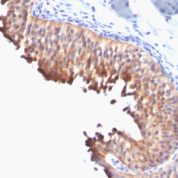
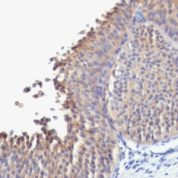
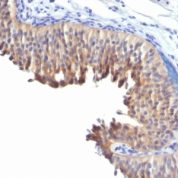
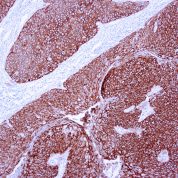
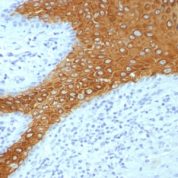
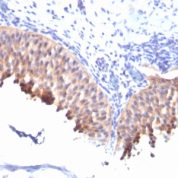
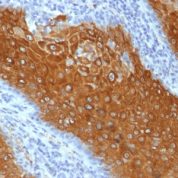
Recommended Positive Control Sample: Skin
Cellular Localization of Antibody DE-K10 Staining: Cytoplasmic
Buffer and Stabilizer: PBS with 1% BSA and 0.05% NaN3
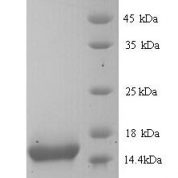
Antibody Concentration: Lot specific. Plese contact tech support for data.
Immunogen: BALB/C mice were immunized with detergent insoluble extract of human epidermis.
Storage Conditions: This antibody should be stored refrigerated (2-8°C). This product should not be used past the expiration date printed on the vial.
Cytokeratin 10 Information for Pathologists
Summary:
Molecular weight of 56.5 kDa. Partner of CK1. CK1 and CK10 are present in suprabasal terminally differentiating cells. Mutations in CK10 or CK1 cause epidermolytic hyperkeratosis / bullous congenital ichthyosiform erythroderma of Brocq (Hum Mol Genet 2006;15:1133, Dermatol Online J 2006;12:6); defects of CK10-CK1 protein network cause structural instability and weakness of keratinocytes, causing blistering, hyperproliferation and hyperkeratosis. CK10 is putative autoantigen in chronic, antibiotic resistant Lyme arthritis (J Immunol 2006;177:2486).Common Uses By Pathologists:
Helps distinguish inflammatory linear verrucous epidermal nevus (higher CK10) from psoriasis (Eur J Dermatol 2004;14:216). Increase indicates a response to treatment for psoriasis (J Am Acad Dermatol 2004;51:257). Positive staining - normal Epidermal spinous and granular cell layers. Positive staining - diseaseLimitations and Warranty
This antibody is manufactured in accordance with clinical good manufacturing practices in an ISO13485:2016 certified production facility. It is intended for multiple uses including in vitro diagnostic use and research use only applications. Please see vial label for expiration date. We strive to always deliver antibodies with a shelf life of at least two years.




There are no reviews yet.