Antibody (Suitable for clinical applications)
Sample Type: FFPE Patient Samples.
Tested Applications: IHC. Approved for In Vitro Diagnostic Procedures on FFPE tissues. For tissue collection recommendations, please see datasheet sent with product.
Application Notes
| Specification | Recommendation |
|---|---|
| Recommended Dilution (Conc) | 1:50-1:100 |
| Pretreatment | EDTA Buffer pH 8.0 |
| Incubation Parameters | 30 min at Room Temperature |
Prior to use, inspect vial for the presence of any precipitate or other unusual physical properties. These can indicate that the antibody has degraded and is no longer suitable for patient samples. Please run positive and negative controls simultaneously with all patient samples to account and control for errors in laboratory procedure. Use of methods or materials not recommended by enQuire Bio including change to dilution range and detection system should be routinely validated by the user.
Clonality: Monoclonal
Anti-Cytokeratin 20 Antibody Clone: EP23
Host and Isotype: Rabbit Rabbit IgG
Recommended Positive Control Sample: Colon
Cellular Localization of Antibody EP23 Staining: Cytoplasmic
Buffer and Stabilizer: PBS with 1% BSA and 0.05% NaN3
Antibody Concentration: Lot specific. Plese contact tech support for data.
Immunogen: A synthetic peptide corresponding to residues near the C-term of human CK20 protein
Storage Conditions: This antibody should be stored refrigerated (2-8°C). This product should not be used past the expiration date printed on the vial.
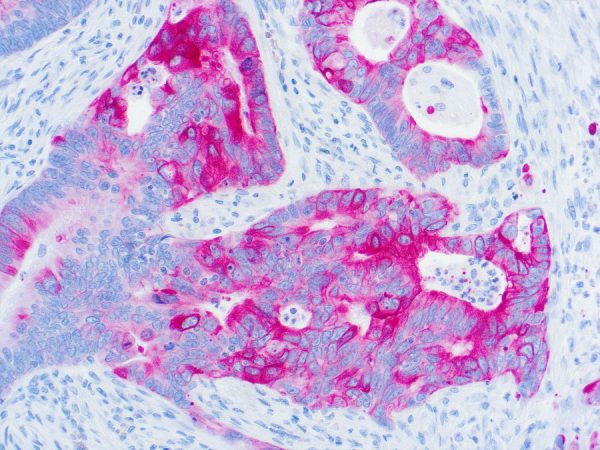
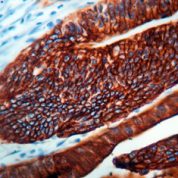
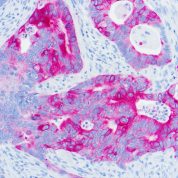
Cytokeratin 20 Information for Pathologists
Summary:
Epithelial marker (MW 46 kDa) with restricted expression compared to CK7 (OMIM #608218). Uses by pathologists General patterns (specificity varies). CK7+ / CK20+ in carcinomas of bile duct (extrahepatic / gallbladder, often, Pathol Res Pract 2003;199:65), lung-mucinous bronchioloalveolar (Am J Clin Pathol 2004;122:421), pancreas (Cancer 2006;106:693, but see Arch Pathol Lab Med 2000;124:1196). Urothelium (often, Arch Pathol Lab Med 2001;125:921, Hum Pathol 2002;33:1136).Common Uses By Pathologists:
General patterns (specificity varies). CK7+ / CK20+ in carcinomas of bile duct (extrahepatic / gallbladder, often, Pathol Res Pract 2003;199:65), lung-mucinous bronchioloalveolar (Am J Clin Pathol 2004;122:421), pancreas (Cancer 2006;106:693, but see Arch Pathol Lab Med 2000;124:1196). Urothelium (often, Arch Pathol Lab Med 2001;125:921, Hum Pathol 2002;33:1136). Also primary mucinous tumors of ovary (74%), upper GI tract (78%) and endocervix (88%, Am J Surg Pathol 2006;30:1130). CK7+ / CK20- in carcinomas of bile duct (intrahepatic, Pathol Res Pract 2003;199:65), breast (Ann Diagn Pathol 1999;3:350), endocervical and endometrial adenocarcinoma (Pathol Res Pract 2003;199:65), esophagus (distal, Am J Surg Pathol 2002;26:1213), lung (not mucinous bronchioloalveolar, BMC Cancer 2006;6:31), salivary gland (Pathol Int 2005;55:386), thyroid (Appl Immunohistochem Mol Morphol 2000;8:189).Limitations and Warranty
This antibody is manufactured in accordance with clinical good manufacturing practices in an ISO13485:2016 certified production facility. It is intended for multiple uses including in vitro diagnostic use and research use only applications. Please see vial label for expiration date. We strive to always deliver antibodies with a shelf life of at least two years.




There are no reviews yet.