Human Anti-EGFR Antibody Product Attributes
Species: Human
Tested Applications: Flow Cytometry, Immunofluorescence, Immunohistochemistry (IHC).
Application Notes: Flow Cytometry (0.5-1ug of antibody/million cells in 0.1ml), Immunofluorescence (1-2ug of antibody/ml), Immunohistochemistry (IHC) (Formalin-fixed) (2-4ug of antibody/ml for 30 minutes at RT)
Clonality: Monoclonal
Anti-EGFR Antibody Clone: GFR1195
Clone GFR1195 Host and Isotype: Mouse IgG1 kappa
Anti-Human EGFR Positive Control Sample: A431 cells. Placenta, Breast, Colon or Bladder cancer
Cellular Localization of Antibody Cell surface
Buffer and Stabilizer: 10mM PBS with 0.05% BSA & 0.05% azide.
Antibody Concentration: 200ug/ml
Antibody Purification Method:Protein A/G Purified
Immunogen: Recombinant human EGFR protein
Storage Conditions: Store at 2 to 8° C (refrigerate). Stable for 24 months when properly stored.
EGFR Previously Observed Antibody Staining Patterns
Observed Subcellular, Organelle Specific Staining Data:
Anti-EGFR antibody staining is expected to be primarily localized to the plasma membrane.Observed Antibody Staining Data By Tissue Type:
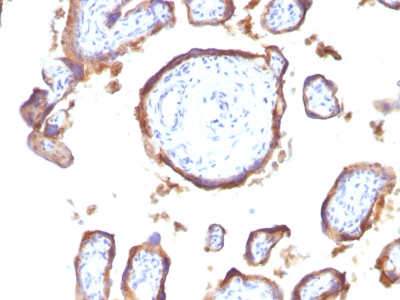
Variations in EGFR antibody staining intensity in immunohistochemistry on tissue sections are present across different anatomical locations. An intense signal was observed in trophoblastic cells in the placenta. More moderate antibody staining intensity was present in trophoblastic cells in the placenta. Low, but measureable presence of EGFR could be seen inhepatocytes in liver, Leydig cells in the testis, melanocytes in skin, myocytes in skeletal muscle, squamous epithelial cells in the tonsil and vagina and urothelial cells in the urinary bladder. We were unable to detect EGFR in other tissues. Disease states, inflammation, and other physiological changes can have a substantial impact on antibody staining patterns. These measurements were all taken in tissues deemed normal or from patients without known disease.Observed Antibody Staining Data By Tissue Disease Status:
Tissues from cancer patients, for instance, have their own distinct pattern of EGFR expression as measured by anti-EGFR antibody immunohistochemical staining. The average level of expression by tumor is summarized in the table below. The variability row represents patient to patient variability in IHC staining.| Sample Type | breast cancer | carcinoid | cervical cancer | colorectal cancer | endometrial cancer | glioma | head and neck cancer | liver cancer | lung cancer | lymphoma | melanoma | ovarian cancer | pancreatic cancer | prostate cancer | renal cancer | skin cancer | stomach cancer | testicular cancer | thyroid cancer | urothelial cancer |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal Intensity | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| EGFR Variability | + | + | + | + | + | ++ | + | ++ | + | + | + | + | + | + | + | + | + | + | + | + |
Limitations and Warranty
enQuire Bio's EGFR Anti-Human Monoclonal is available for Research Use Only. This antibody is guaranteed to work for a period of two years when properly stored.



-178x178.jpg)
![Analysis of Mass Spec data (dashed-line) of fractions stained with EGFR MS-QAVA™ monoclonal antibody [Clone: H9B4] (solid-line), reveals that less than 13.1% of signal is attributable to non-specific binding of anti-EGFR [Clone H9B4] to targets other than EGFR protein. Even frequently cited antibodies have much greater non-specific interactions, averaging over 30%. Data in image is from analysis in Jurkat, U202 and HeLa cells.](https://cdn-enquirebio.pressidium.com/wp-content/uploads/2017/10/enQuire-Bio-1956-MSM3-P1-anti-EGFR-antibody-178x178.png)

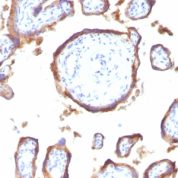
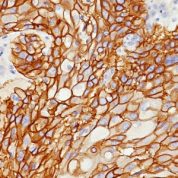
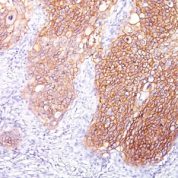
![Staining with mouse monoclonal EGFR [Clone 31G7] antibody in formalin-fixed paraffin-embedded human placenta.](https://enquirebio.com/wp-content/uploads/2019/01/EGFR IHC human Placenta (31G7)-178x178.jpg)
There are no reviews yet.