Human and Horse Anti-Renal Cell Carcinoma / gp200 Antibody Product Attributes
Species: Human and Horse
Tested Applications: Flow Cytometry, Immunofluorescence, Western Blot, Immunohistochemistry (IHC).
Application Notes: Flow Cytometry (0.5-1ug of antibody/million cells in 0.1ml), Immunofluorescence (1-2ug of antibody/ml), Western Blot (0.5-1ug of antibody/ml), Immunohistochemistry (IHC) (Formalin-fixed) (0.5-1ug of antibody/ml for 30 minutes at RT)
Clonality: Monoclonal
Anti-Renal Cell Carcinoma / gp200 Antibody Clone: 66.4.C2
Clone 66.4.C2 Host and Isotype: Mouse IgG2b kappa
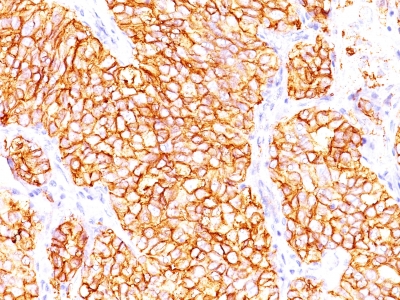
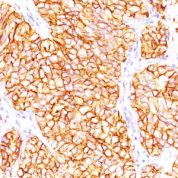
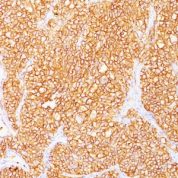
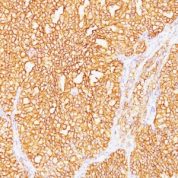
Anti-Human and Horse Renal Cell Carcinoma / gp200 Positive Control Sample: Normal kidney or renal cell carcinoma
Cellular Localization of Antibody <66.4.C2 Staining: Cell surface, cytoplasmic
Buffer and Stabilizer: 10mM PBS with 0.05% BSA & 0.05% azide.
Antibody Concentration: 200ug/ml
Antibody Purification Method:Protein A/G Purified
Immunogen: Microsomal fraction of human renal cortical tissue homogenate
Storage Conditions: Store at 2 to 8° C (refrigerate). Stable for 24 months when properly stored.
Renal Cell Carcinoma / gp200 Previously Observed Antibody Staining Patterns
Observed Subcellular, Organelle Specific Staining Data:
Anti-CA9 antibody staining is expected to be primarily localized to the plasma membrane and cytosol.Observed Antibody Staining Data By Tissue Type:
Variations in Renal Cell Carcinoma / gp200 antibody staining intensity in immunohistochemistry on tissue sections are present across different anatomical locations. An intense signal was observed in glandular cells in the gallbladder, stomach. More moderate antibody staining intensity was present in glandular cells in the gallbladder, stomach. Low, but measureable presence of Renal Cell Carcinoma / gp200 could be seen in. We were unable to detect Renal Cell Carcinoma / gp200 in other tissues. Disease states, inflammation, and other physiological changes can have a substantial impact on antibody staining patterns. These measurements were all taken in tissues deemed normal or from patients without known disease.Observed Antibody Staining Data By Tissue Disease Status:
Tissues from cancer patients, for instance, have their own distinct pattern of Renal Cell Carcinoma / gp200 expression as measured by anti-Renal Cell Carcinoma / gp200 antibody immunohistochemical staining. The average level of expression by tumor is summarized in the table below. The variability row represents patient to patient variability in IHC staining.| Sample Type | breast cancer | carcinoid | cervical cancer | colorectal cancer | endometrial cancer | glioma | head and neck cancer | liver cancer | lung cancer | lymphoma | melanoma | ovarian cancer | pancreatic cancer | prostate cancer | renal cancer | skin cancer | stomach cancer | testicular cancer | thyroid cancer | urothelial cancer |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal Intensity | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| CA9 Variability | + | + | + | + | + | + | + | + | + | + | + | + | + | + | ++ | + | ++ | + | + | ++ |
Limitations and Warranty
enQuire Bio's Renal Cell Carcinoma / gp200 Anti-Human, Horse Monoclonal is available for Research Use Only. This antibody is guaranteed to work for a period of two years when properly stored.



There are no reviews yet.