Antibody (Suitable for clinical applications)
| Specification | Recommendation |
|---|---|
| Recommended Dilution (Conc) | 1:25-1:50 |
| Pretreatment | Citrate Buffer pH 6.0 |
| Incubation Parameters | 30 min at Room Temperature |
Prior to use, inspect vial for the presence of any precipitate or other unusual physical properties. These can indicate that the antibody has degraded and is no longer suitable for patient samples. Please run positive and negative controls simultaneously with all patient samples to account and control for errors in laboratory procedure. Use of methods or materials not recommended by enQuire Bio including change to dilution range and detection system should be routinely validated by the user.
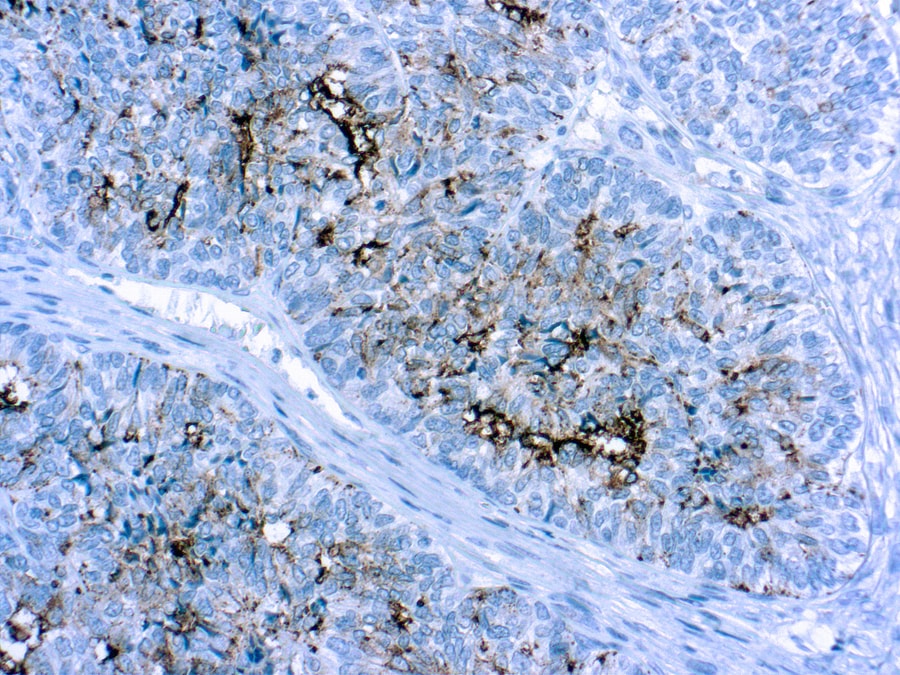
CA 125 Information for Pathologists
Summary:
Cancer antigen 125 (CA-125) is a protein encoded by MUC16 gene, used as a tumor marker, because it is 79% sensitive for ovarian cancer. Not specific, because elevated in other tumors and in inflammatory conditions. Terminology Also known as MUC16. Pathophysiology
Notable Clinical Features:
Elevated in carcinomas of breast, cervix (Asian Pac J Cancer Prev 2010;11:1745), endometrium, fallopian tubes, GI, lung, ovary; also endometriosis, pregnancy, inflammatory disorders. Interpretation Strong membranous staining, fallopian tube is good control. Uses by pathologists (1) For ovarian carcinomas, use serum levels to follow response to treatment and predict prognosis (Ann Oncol 2008;19:327).
Common Uses By Pathologists:
(1) For ovarian carcinomas, use serum levels to follow response to treatment and predict prognosis (Ann Oncol 2008;19:327). (2) In UK, recommended to use serum levels to screen women with symptoms of ovarian cancer (National Health Service Press Release). (3) Serial measurements may be more specific for ovarian cancer. (4) Useful in immunohistochemistry to confirm ovarian origin of tumor (Am J Surg Pathol 2005;29:1482). (5) Serum levels may have role in assessing heart failure (Clin Cardiol 2011;34:244, Eur Heart J 2010;31:1752).
| CA 125 General Information | |
|---|---|
| Alternate Names | |
| Molecular Weight | |
| 1519.2 kDa | |
| Chromosomal Location | |
| p13.2 [chr: 19] [chr_start: 8848844] [chr_end: 8981342] [strand: -1] | |
| Curated Database and Bioinformatic Data | |
| Gene Symbol | MUC16 |
| Entrez Gene ID | 94025 |
| RefSeq Protein Accession(s) | NP_078966 |
| RefSeq mRNA Accession(s) | XM_017027489; XM_017027487; XM_017027499; XM_017027500; XM_017027486; XM_017027493; NM_024690; XM_017027495; XM_017027488; XM_017027490; XM_017027494; XM_017027501; XM_017027491; XM_017027492 |
| RefSeq Genomic Accession(s) | NC_000019; NG_055257 |
| UniProt ID(s) | Q8WXI7 |
| PharmGKB ID(s) | PA31314 |
| KEGG Gene ID(s) | hsa:94025 |
| General Description of CA 125 . | |
| This antibody reacts specifically with CA 125 ovarian cancer antigen. Several studies have shown that CA 125 is a useful tumor marker for ovarian epithelial malignancies. However, CA 125 has been described in other neoplasms such as seminal vesicle and anaplastic lymphomas. | |



Reviews
There are no reviews yet.